Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 C based on two binary non-ideal solid solutions
C based on two binary non-ideal solid solutionsWalker, C.; Suto, Shunkichi; Oda, Chie; Mihara, Morihiro; Honda, Akira
Cement and Concrete Research, 79, p.1 - 30, 2016/01
Times Cited Count:69 Percentile:90.42(Construction & Building Technology)Modeling the solubility behavior of calcium silicate hydrate (C-S-H) gel is important to make quantitative predictions of the degradation of hydrated ordinary Portland cement (OPC) based materials. Experimental C-S-H gel solubility data have been compiled from the literature, critically evaluated and supplemented with new data from the current study for molar Ca/Si ratios = 0.2-0.83. All these data have been used to derive a discrete solid phase (DSP) type C-S-H gel solubility model based on two binary non-ideal solid solutions in aqueous solution(SSAS). Features of the DSP type C-S-H gel solubility model include satisfactory predictions of pH values and Ca and Si concentrations for all molar Ca/Si ratios = 2.7  0 in the C-S-H system, portlandite (CH) for Ca/Si ratios
0 in the C-S-H system, portlandite (CH) for Ca/Si ratios  1.65, congruent dissolution at Ca/Si ratios = 0.85, and amorphous silica (SiO
1.65, congruent dissolution at Ca/Si ratios = 0.85, and amorphous silica (SiO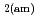 ) for Ca/Si ratios
) for Ca/Si ratios  0.55 as identified in the current study by IR spectroscopy.
0.55 as identified in the current study by IR spectroscopy.
Suzuki, Hiroshi; Bae, S.*; Kanematsu, Manabu*
Advances in Materials Science and Engineering, 2016, p.8936084_1 - 8936084_6, 2016/00
Times Cited Count:2 Percentile:7.52(Materials Science, Multidisciplinary)The deformation behavior of nanostructure of calcium silicate hydrate (CSH) in Portland cement (PC) paste under compression was successfully characterized by the atomic pair distribution function (PDF) measured by using Synchrotron X-rays. The PDF of the PC paste showed a unique deformation behavior for a short range order below 2.0 nm in radius corresponding to the size of the CSH particle (globule), while the deformation for a long range order was similar to that of a calcium hydroxide phase measured by the diffraction peak shift. The compressive deformation of the CSH nanostructure can be divided into three stages with different interactions between globules. This behavior would originate from the granular nature of CSH which deforms with increasing packing density by slipping the interfaces between globules, rearranging the overall CSH nanostructure. This study will lead to increasing applications of the PDF technique to provide clues for understanding the deformation mechanism of CSH in PC paste.
Maeda, Toshikatsu; Bamba, Tsunetaka*; Hotta, Katsutoshi*; Mizuno, Tsuyoshi*; Ozawa, Tatsuya
Nihon Genshiryoku Gakkai Wabun Rombunshi, 4(4), p.242 - 247, 2005/12
no abstracts in English
Suzuki, Hiroshi; Kanematsu, Manabu*; Bae, S.*; Shiro, Ayumi; Shobu, Takahisa
no journal, ,
The deformation behavior of nanostructure of calcium silicate hydrate (CSH) in Portland cement (PC) paste under compression was successfully characterized by the atomic pair distribution function (PDF) measured by using Synchrotron X-rays. The PDF of the PC paste showed a unique deformation behavior for a short range order below 2.0 nm in radius corresponding to the size of the CSH particle (globule), while the deformation for a long range order was similar to that of a calcium hydroxide phase measured by the diffraction peak shift. The compressive deformation of the CSH nanostructure can be divided into three stages with different interactions between globules. This behavior would originate from the granular nature of CSH which deforms with increasing packing density by slipping the interfaces between globules, rearranging the overall CSH nanostructure. This study will lead to increasing applications of the PDF technique to provide clues for understanding the deformation mechanism of CSH in PC paste.
Rizaal, M.; Nakajima, Kunihisa; Osaka, Masahiko; Saito, Takumi*; Erkan, N.*; Okamoto, Koji*
no journal, ,
Experimental study using thermogravimetry and X-ray diffractometry was conducted to investigate chemical interaction between calcium silicate insulation and cesium hydroxide under steam and hydrogen atmospheres. This interaction might be one of the causes for the high dose rate reading outside the pedestal in 1F, Unit2. Our results suggested that some chemical interaction occurred in temperature range of 575-730 C regardless of atmospheres.
C regardless of atmospheres.